Yury Miller Lab
- Current Projects
- Past Projects
- Lab Members
- News
- Lab Alumni
Atherosclerosis PAin Alzheimer’s Asthma
The excessive accumulation of cholesterol in vascular macrophages is regarded as a leading factor in the development of vascular inflammation, plaque instability and clinical manifestations of atherosclerosis. However, inflammatory genes are predominantly expressed in macrophages that accumulate cholesterol in the plasma membrane rather than in lipid droplet-laden macrophage foam cells. Cholesterol and many receptors governing inflammatory responses colocalize in the ordered plasma membrane microdomains, often designated as lipid rafts. Upon activation, lipid raft resident and recruited proteins assemble and initiate signaling cascades leading to inflammation. We introduced the term inflammarafts, defined as enlarged, clustered lipid rafts harboring activated receptors and adaptor molecules and serving as a scaffold to organize cellular inflammatory responses. We found inflammarafts to be surprisingly stable in macrophages isolated from atherosclerotic lesions. We further identified apoA-I binding protein (AIBP) as a key regulator of cellular cholesterol metabolism, which can selectively target inflammarafts via its binding to TLR4, without disrupting physiological lipid rafts. Hypercholesterolemic AIBP deficient mice, which we created, developed exacerbated atherosclerosis. In contrast, the AAV-mediated expression of a secreted form of AIBP in the liver reduced atherosclerosis. In addition, mitochondria in AIBP-deficient cells were morphologically distorted, with a characteristic hyper-branched and cupped shape, typically associated with oxidative stress. The goal of this project is to delineate mechanisms governing differential inflammaraft dynamics and related mitochondrial dysfunction in macrophage foam cells and in non-foamy macrophages in atherosclerosis.

Persistent pain secondary to tissue and nerve injury involves key roles for dorsal root ganglia (DRG) neurons, macrophages, and spinal microglia. Therapies targeting single components of this tripartite pain axis have not proven clinically efficacious, suggesting a strategy is needed that addresses all three components in concert. We have exciting new findings that suggest one such therapeutic strategy may be to target the subset of cellular membrane cholesterol-rich lipid rafts that contain Toll-like receptor 4 (TLR4-rafts). Membrane lipid rafts are a fundamental organizational nidus in all cells for numerous membrane channels, receptors and enzymes that regulate reactivity and excitability. The TLR4-raft subtype is special as these are specifically localized in the three key cellular elements regulating the excitability of nociceptive signaling. Following tissue inflammation or nerve injury, TLR4-rafts transform from small diffuse labile membrane structures into markedly enlarged and persistent complexes supporting the formation of homo and hetero dimers of several excitatory channels and receptors, underpinning the transition from an acute to a persistent pain phenotype and to establishing a “primed state” after an initial system activation. Specific disruption of these neuraxial TLR4-rafts has profound effects upon injury induced pain behavior. This project will have a sustained impact on understanding the neurobiology of a largely unexplored cellular element regulating post injury afferent excitability; and point to the unifying thesis that neuraxial TLR4-rafts are a common facilitatory element of the persistent pain facilitated states observed following tissue and nerve injury and mediated by DRG nociceptors, macrophages, and spinal microglia.

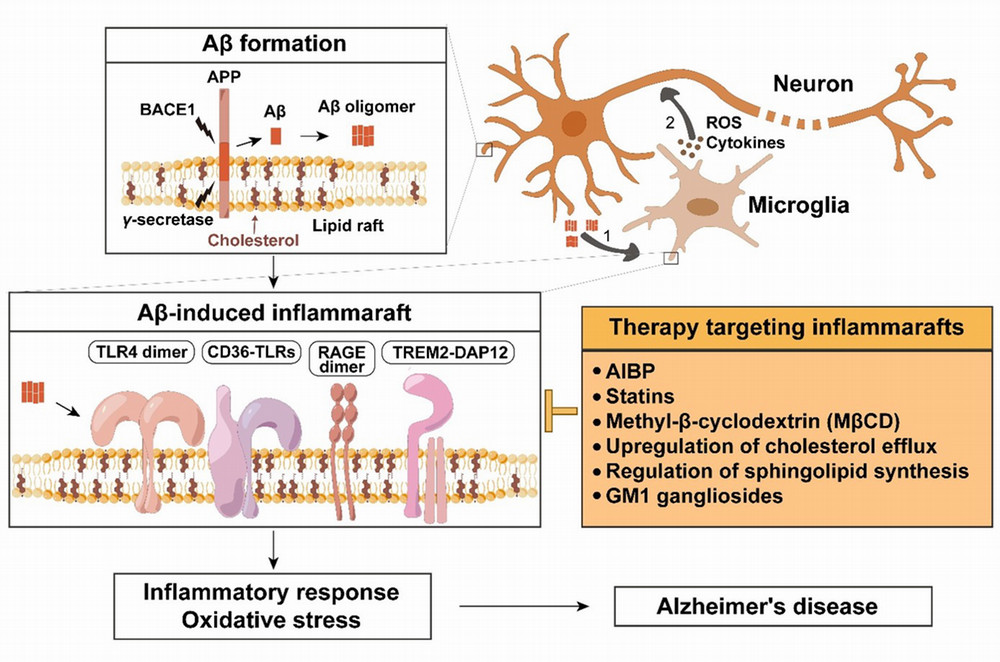
Neuroinflammation is a major factor in the progression of Alzheimer’s disease (AD). Inflammatory brain microglia are characterized by altered cholesterol and lipid metabolism. Cholesterol and many receptors governing inflammatory responses colocalize in the ordered membrane microdomains often designated as lipid rafts. Upon activation, lipid raft resident and recruited molecules assemble and initiate signaling cascades leading to inflammation. We have identified the apoA-I binding protein (AIBP, encoded by the APOA1BP gene) as a key regulator of cellular cholesterol metabolism, which can selectively target lipid rafts in inflammatory cells via its binding to TLR4. While extracellular AIBP regulates cholesterol depletion from the plasma membrane and controls lipid rafts, intracellular AIBP localizes to mitochondria, facilitates mitophagy and helps maintain normal mitochondrial function and control oxidative stress. Apoa1bp-/- APP/PS1 mice present more amyloid beta (Aβ) plaques, an exacerbated dysfunctional microglia phenotype and show increases in cell death when compared to APP/PS1 mice. Mitochondria in AIBP-deficient microglia are morphologically distorted, with a characteristic hyper-branched and cupped shape, typically seen following oxidative stress. The goal of this project is to delineate mechanisms governing protective effects of AIBP in the AD brain, focusing on microglial lipid rafts and on mitochondrial dysfunction.
Hypersensitivity to the common cold virus rhinovirus (RV) triggers severe, often fatal exacerbations in asthmatics. Poorly managed aspects of severe asthma include a strong inflammatory component, particularly following viral infection. Bronchial epithelial cells (BECs) are the first-responder cells that produce alarmins to initiate the immune response. When exposed to RV, BECs also produce, in a TLR2-dependent manner, neutrophil chemoattractants. Excessive neutrophilia is a major factor in RV-induced severe asthma exacerbation. This project is to test the hypothesis that exposure to allergens reprograms BECs to chronically express stable inflammarafts, defined as enlarged, cholesterol-rich lipid rafts hosting assembled complexes of inflammatory receptors tuned up for production of excessive immune response to microbial infection. We discovered that in chronic inflammation, many cell types express inflammarafts for as long as weeks and even months. We demonstrated that house dust mite (HDM), a common allergen in the pathogenesis of asthma, metabolically reprograms BECs to express inflammarafts enriched with TLR2, the receptor that recognizes RV and mediates expression of neutrophil chemoattractants. The molecular mechanisms identified in this project will help develop novel therapeutic strategies to reverse the inflammaraft reprogramming of BECs and reduce hypersensitivity to viral infection in severe asthma.